Remember those vintage chemistry sets that we opened up the other day? We left out one of the coolest things that came along with them: A thick stack of fantastic manuals, instructions, posters, and charts. Let’s take a look through them!

First off, the thickest manual in the stack. The cover is titled “Adventures With Chemistry,” and the title page inside reads “The Master Chemcraft Manual” by Harold M Porter, copyright 1951. Harold Porter was the owner of the Porter Chemical Company, which sold over a million chemistry sets from the 1950’s to 1980’s.
Arguably, then, this book was a best seller. And with good reason, too: it really is a “master manual,” with 716 clearly-described experiments in 230 pages. (And it deserves to go back into print.)

Some of the experiments are the ones you’d expect to find in a chemistry set, requiring test tubes of chemicals and an alcohol burner.

Other experiments are of the “kitchen science” sort– like separating gluten from wheat or preparing the milk protein casein.

This is the manual for the excellent handheld spectroscope that came in some of the chemistry sets.
If only this chart were in the same vibrant color from the front page of the manual!
(You may find this chart to be a helpful modern substitute.)

The illustration shows a young chemist looking through a spectrometer at the flame produced by a sample of sodium bisulfate, as he blows and torches it with a handheld “blow torch” of the traditional sort: An alcohol burner with a mouth-blown tube.
Conclusion: chemistry was considerably awesomer back in the 1950’s!
The Lionel Outer Space Manual, (inside title: “Conquering Outer Space Through Chemistry”). It’s easy to imagine getting this booklet, with the expectation that it will tell you how to build your own space capsule and visit the moon.
Excellent; there will be room for passengers, too!
However, to everyone’s great disappointment, the rocket that you actually get to build involves a soda bottle, baking soda, a cork, and round pencils for bearings (so that it can roll a little bit).
The Atomic Energy manual is heavy on storytelling and light on number of experiments, but the few experiments described are indeed safe and exciting.
“Uranium is, of course, entirely safe to handle as its radioactivity is relatively low.” That is still true today, but it should be followed up with “but wash your hands thoroughly with soap and warm water after handling it.”
Somehow radium didn’t make it into our charts on the monetary density of things. But at a modern price of 40 M$/Lb, it is still (right up there with LSD) one of the most valuable substances in the world.
“Let’s Explore Chemistry” by Nathan Feifer, copyright 1959. This is an actual book, and not one that came with a chemistry set. And while it is charming, it has a much lower density of experiments than Adventures With Chemistry.
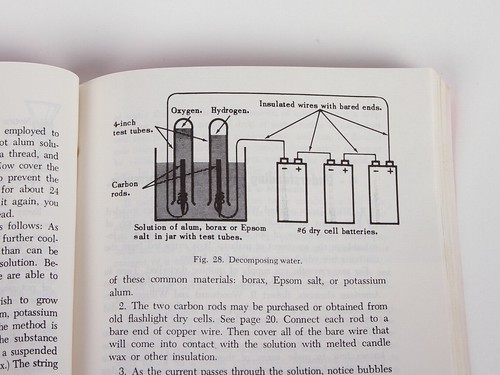
Amongst other experiments, it shows you how to decompose water by electrolysis; a classic, fun experiment that you should perform if you haven’t already. Unfortunately, batteries no longer come with convenient carbon rods in the middle. (Perhaps thick pencil leads would work well.) There are a lot of other experiments described here as well, some of which are much less interesting.
Next up: A freaking glass blowing manual!
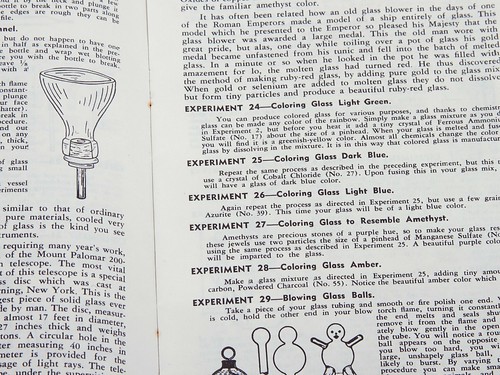
This is really something, full of instructions on how to color, blow, and join glass. (And something that you’d never find in a manual for young teens today!)
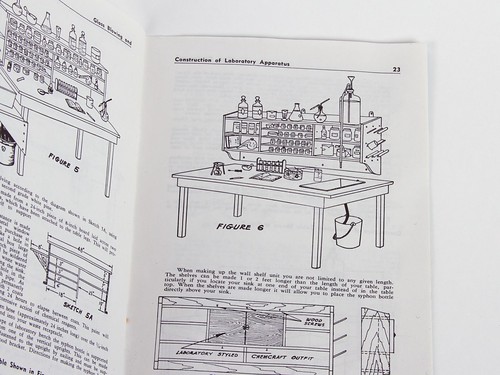
It’s all working up to building a full-on chemistry lab bench at home.
“Your World of SCIENCE,” which has this wonderful cover and… not much else. The inside consists of 16 short pages of general science facts and tepid experiments. Plus a coupon to join the Chemcraft Science Club!
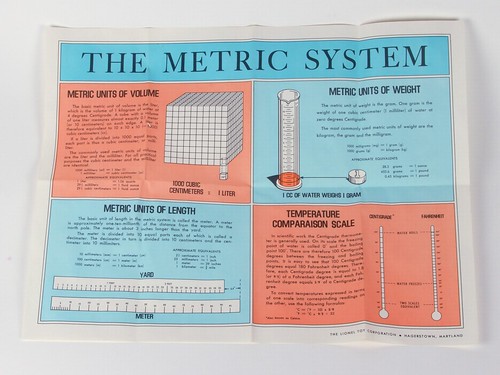
There were two folded-up poster charts in the stack as well: An introduction to the metric system and an old-style periodic table that goes up to 98.
This little manual doesn’t look like much, but it’s actually one of my favorite parts, full of detailed instructions about using the scale, hydrometer, centrifuge, and agitator accessories that came with the chemistry set.
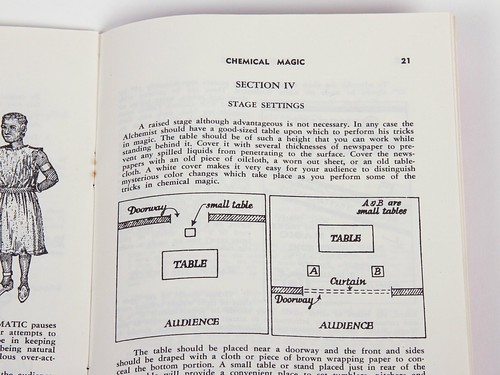
And then there’s what is unquestionably the strangest and most dated of the manuals: the “Chemical Magic” manual, complete with how to set your stage for performing chemistry tricks.
It even has a (genuinely cringe-inducing) script to read as you perform some of the 90-odd flashy experiments listed in the booklet.
And finally, “The Famous Chemcraft Charts”: a set of tiny looseleaf wall charts.
The metric system of weights and measures. The pH of solutions. The periodic table of the elements. Specific gravity.
Measurement of temperature. Metric units of length. Metric units of volume. And a table of the elements, their symbols, atomic weights and numbers.
If you look at that last chart a little more closely, you might notice a few differences from the modern table:
Not only does the table only go up to element 101, but Niobium had symbol Cb, and Einsteinium symbol E. Both were correct at the time the chart was published; the symbols have changed since then (to Nb and Es, respectively). Our modern table has named elements as high as 116, Livermorium, with symbol Lv.































Spiffy! And apparently still commercially available: http://www.essex1.com/people/speer/chem.html
I have not contacted this vendor; it is only internet research.
I’m a member of a radiation enthusiasts group, largely centered around geiger counters. A few years ago I learned about Spinthariscopes (Victorian TV) and found how to make one easily. You need a radium watch hand and a $3 loupe, and you need to be dark-adapted. Some were even built and sold on eBay by other hobbyists.
Do be careful with radium paint it may flake and it does emit radon. Not all ‘luminous’ hands are radium, they have to be older.
Brings back fun memories. (Especially the keep out reach of children labels) Through Hazmat training I have recently learned that some vintage Chem sets contain Pitric Acid, which can become unstable and dangerous if it dries out. Always check list of chemicals before shaking it too much. Check out gov safety site below.
http://oag.ca.gov/sites/all/files/pdfs/cci/safety/picric.pdf
When I was a kid, I went to the local chemistry/biology supply house and requested all of the chemicals on my (relatively short) shopping list to add to my chemistry set. The *one* that they wouldn’t sell me was picric acid. Wasn’t clear if that’s because they didn’t have it, or because they didn’t think that I should have it. But, lesson learned.
The real classic home chemistry manual, though, is “The Golden Book of Chemistry Experiments” by Robert Brent with illustrations by Harry Lazurus. See Marie and Pierre Curie gaze with wonder at a gram of glowing radium extracted from tons of coal, watch Kekule dream of a snake eating its tail (benzene structure), set up your home lab and generate chlorine gas to amaze your friends. This book was probably responsible for a whole generation of kids like me who ended up becoming chemists.
You can download it here:
http://chemistry.about.com/library/goldenchem.pdf
Download it here: http://chemistry.about.com/b/2012/10/01/download-the-golden-book-of-chemistry-experiments.htm
It’s the glassblowing book (and indeed the equipment to for it) that I would like. I always felt that a real chemist should be able to blow glass!